Jual Reagen PCR – Pandemi virus corona yang cepat membutuhkan penanganan taktis dari kita semua. Disamping membatasi kegiatan di luar rumah dengan PSBB(Pembatasan Sosial Bersekala Besar), kita juga perlu melakukan pemeriksaan spesimen dalam jumlah yang cukup banyak. Rapid Test dan Real Time PCR merupakan metode yang digunakan untuk menguji apakah seseorang terjangkiti virus corona atau tidak.
PT. Andaru Persada Mandiri sebagai distributor alat laboratorium selain menyediakan alat Real Time PCR juga jual reagen PCR untuk kebutuhan uji COVID-19. Jika anda membutuhkan reagen untuk uji corona virus, silahkan hubungi kami di WhatsApp : +6287777277740



Spesifikasi Reagen PCR
Berikut ini adalah spesifikasi teknis dari reagen yang bisa digunakan untuk uji corona virus. Beberapa diantara anda mungkin merasa bingung dan belum mengerti, apakah reagen yang kami sediakan compatible(bisa digunakan) dengan Real Time PCR yang telah mereka miliki. Jika anda mengalami tersebut, silahkan hubungi kontak kami atau konsultasi via WhatsApp : +6287777277740
Novel Coronavirus(2019-nCoV) Nucleic Acid Diagnostic Kit (PCR-Fluorescence Probing) is used for qualitative detection of the ORF1ab and N genes of novel coronavirus (2019-nCoV) in nasopharyngeal swab, oropharyngeal swab, alveolar lavage fluid, sputum, serum, whole blood and feces from suspected pneumonia cases with novel coronavirus infection, patients with suspected clusters ofnovelcoronavirus infection, and otherpatients requiring diagnosisordifferentialdiagnosisofnovelcoronavirus infection.
Note : For in vitro diagnostic use only. For professional use only.
The definitions of “suspected cases” and “suspected clusters of patients” shall be defined by referring to the “Pneumonia Diagnosis and Treatment Program for Novel Coronavirus Infection” and “Pneumonia Case Monitoring Program for Novel Coronavirus infection” issued by China CDC (the current version).
Novel Coronavirus(2019-nCoV) NucleicAcid Diagnostic Kit (PCR-Fluorescence Probing) is only used for the auxiliary diagnosis of related cases and the emergency reserve for in vitro diagnosis during the pneumonia outbreak of novel Coronavirus (2019-nCoV) infection since December 2019, this kit shouldn’t be used as routine in vitro diagnostic in clinical practice. Please follow the relevant requirements of the “Pneumonia Diagnosis and Treatment Program for Novel Coronavirus Infection”, “Pneumonia Prevention and Control Program for Novel Coronavirus infection” and other documents in use.
The novel Coronavirus nucleic acid tests should comply with “Technical Guidelines for Laboratory Testing of Novel Coronavirus” and keep good biosafety.
By applying Real-time fluorescence quantitative RT-PCR technology on the fluorescence quantitative PCR instrument, this test utilizes the novel coronavirus (2019-nCoV) ORF 1ab and the specific conserved sequence of coding nucleocapsid protein N gene as the target regions which are designed for the conserved sequence of the double-target genes, to achieve detection of sample RNAthrough fluorescent signal changes.
The PCR detection system uses the positive internal control, which monitors the presence of PCR inhibitors in test specimens by detecting whether the internal control signal is normal, to avoida falsenegative result.
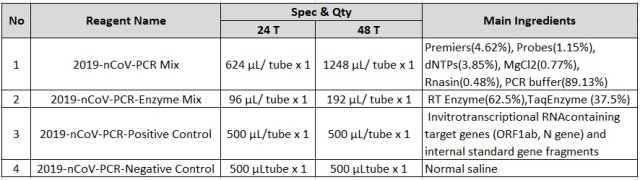
This kit is an amplification reaction reagent and contains the following components:
Note:
1. Do not mix or exchange components from different kit lots.
2. Allbiological samplesin the diagnostic kithave been inactivated.
3. Materials required but not provided: 1.5 mL DNase-free and RNase-free centrifuge tubes, 0.2 mL PCR reaction tubes, pipette tips (10 μL, 200 μL and 1000 μL tips with filters are preferred), desktop centrifuge, desktop vortex mixer various models of pipette guns.
4. Self-prepared reagent: Sample Release Reagent ( Reference Number : S1014E ) or Sample Release Reagent (Reference Number : S1015E) or Nucleic Acid (DNA/RNA) Extraction or Purification Kit (Magnetic beads method) ( Reference Number : S1002E )
1. The diagnostic kit should be stored in a sealed pouch at -20±5°C and protected from light. The kit is provisionally valid for 6 months.
2. Please refer to the date of manufacture and expirydate on the outer package.
3. The reagents keep valid and stable within the expiry date if not used. As long as the container of the reagent is opened, the freeze/thaw cyclesshould not exceed three.
1. Applicable specimen type: nasopharyngeal swab, oropharyngeal swab, alveolar lavage fluid, sputum, serum, whole blood and feces.
2. Collectionof specimen
Nasopharyngeal swab/oropharyngeal swab: Collect sample in accordance with the relevant provisions of “Specimen Collection Method” in the “Pneumonia Laboratory TechnicalGuide for Novel Coronavirus Infection” from “Pneumonia Prevention and Control Plan for Novel Coronavirus Infection”. It is proved that the swab made of nylon sampling head and ABS sampling rod can be selected for sample collection.
Nasopharyngeal swab: The specimen collection tube should be pasted with the barcode first, the nasopharyngeal swab should be collected within 3 days after the onset of the disease as far as possible.Use swab to measure the length between apex nasi and earlobe, then mark with finger. Insert the swab into the nasal cavity in direction of perpendicular to the nose (face). The swab should be inserted at least half of the length from the earlobe to the apex nasi. Make the swab stops in the nasal for 15 ~ 30 s, gently rotate 3 ~ 5 times, quickly put swab into specimen collection tube cotaining 2 mL Lysis Buffer (same as Lysis Buffer in the Sample Release Reagent) or Sample Storage Reagent containing RNA enzyme inhibitor. Insert the swab, then break the sterile swab rod near the top, tighten tube cap and seal withsealing film.
Oropharyngeal swab: The specimen collection tube should be pasted with the barcode first, the oropharyngeal swab should be collected within 3 days after the onset of the disease as far as possible. A sterile flocking swab should be used for sampling, moderately wipe the posterior pharyngeal wall, avoid touching the tongue. Quickly place a sterile swab into the collection tube used for collectionof oropharyngeal swab. Break the sterile swab rod near the top, tighten tube cap and seal with sealing film.
Alveolar lavage fluid: Severe patients or patients with pneumonia who progress rapidly. Clinician extract ≥5 ml BALF into a 50 mL aseptic container labeled with sample bar code and screw cap by aseptic operation. Collect specimen, then tighten tube cap and seal with sealing film.
Sputum: The specimen collectiontube shouldbe pasted with the barcode first.Do not open the airway to collectspecimens when collecting sputum. Collect.deep cough sputum into a disposableaseptic sampling cup withscrew cap, load 2 mLprotease K (1g/L) into sampling cup. Collect sputum, then tighten tube cap and seal with sealing film. Send to detection within 30 min as far as possible. Protease K shouldnot be added first ifspecimens need to be transported over long distances.
Whole blood: Bloodsamplescan be collected within7 days after the onset orcriticalpatients,or patients considered with viremia. The specimen collection tube should be pasted with the barcode first. Collect 2~4 mL of blood samples into vacuum blood collection tube containing EDTA anticoagulant.
Feces: For patients with gastrointestinal symptoms such as diarrhea at the early stage of the disease, preserve 3~5 g (soybean size) feces. The specimen collection tube should be pasted with the barcode first. Collect sample into specimen collection tube with screw cap cotaining 2 mL normal saline (RNAenzyme inhibitor can be added when conditions permit) then seal with sealing film.
After sample collection, itisrecommended to place into Sample Storage Reagent for preservation.
3. Storage and delivery of specimens:
Specimens to be tested can be immediately processed, specimens to be tested within 24 hours can be stored at 4℃. Specimens that cannotbe detected within24 hoursshouldbe stored at-70℃ orbelow(inthe absence of -70℃ storage conditions,specimens to be tested can be stored at -20℃ for 10 days, nucleic acid can be stored at -20±5℃ for 15 days). Multiple freeze/thaw cycles should be avoided. Specimens should be transported in a sealed frozen pitcher with ice or in a sealed foam box with ice. The inactivation of samples at 56℃ for 30min will not affect the detection of this kit.
1. Preparation of reagent (performed at“reagent preparation region”)
1.1 Take out each component from the diagnostickit and place them at room temperature.Allowthe reagents to equilibrate at room temperature, then vortex each of them respectivelyfor later use.
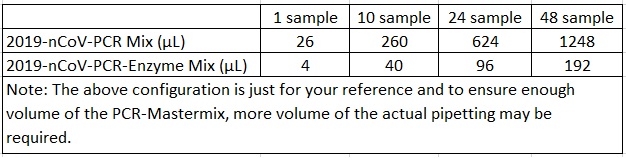
1.2 According to the quantity of test specimens, 2019-nCoV-PCR-Positive Control and 2019-nCoV-PCR-Negative Control, pipette appropriate quantity of 2019-nCoV-PCR Mix and 2019-nCoV-PCR-Enzyme Mix (2019-nCoV-PCR Mix 26 μL/test + 2019-nCoV-PCR-EnzymeMix4μL/test), mixthem thoroughly to makea PCR-Mastermix, centrifuge it instantaneously for later use.
1.3 Transfer the above-prepared reagents to the “specimen processing region” for later use.
2. Processing and loading of specimens (performed at “specimen processing region”)
2.1 Use Sample Release Reagent ( Reference Number : S1014E ), Sample Release Reagent ( Reference Number : S1015E ), NucleicAcid (DNA/RNA) Extraction or Purification Kit (Magnetic beads method) ( Reference Number : S1002E ) manufactured by Sansure Biotech Inc. to extract the nucleic acidas per the product manual.
2.2 Add 30 μLPCR-Mastermixinto PCR reaction tube with 20 μLaboveprocessedsample. Carry out fluorescence quantitative PCR detection on fluorescence PCR instrument. The fluorescent PCR tube can be sealed with 15 μL paraffin oil before PCR amplification.
3. PCRAmplification (Refer to user manual of each instrument to adjust the settings)
3.1 Place PCR reaction tubes into the specimen wells of the amplification equipment. Set up the 2019-nCoV-PCR-Positive Control, 2019-nCoV-PCR-Negative Control and specimens to be tested in the corresponding sequence and input specimen name.
3.2 Select PCR test channel: a) Select FAM (ORF-1ab region) and ROX (N gene) channels to test 2019-nCoV nucleicacid. b) Select CY5 channel to test internal control.
3.3 Set cycle parameters
When the settings are completed, save the settings and carry out the reactionprocedure.
4. ResultAnalysis (Refer to user manual of instrument to adjust the settings.)
Results will be saved automatically when reactions are completed. Analyze amplification curve of target of detection and internal control. Adjust Start, End and Threshold values of Baseline of the graph according to analysis result (Users can adjust the values according to the actual situation. Start value can be set between 3-15, and End value between 5-20.Adjust the amplification curve of negative control to be flat or below threshold). Click “Analyze” to implement the analysis, make sure each parameter satisfy the requirements given in“5. QualityControl”. Go to “Plate” window to record qualitative results.
Diatas adalah beberapa informasi spesifikasi reagen untuk alat Real Time PCR. Beberapa bagian lainnya, seperti :
- Quality Control
- Reference Range
- Explanation of Detection Result
- Limitation of Detection Method
- Product Performa Index, dan
- Precautions
Tidak kami sampaikan karena memang membutuhkan diskusi lebih lanjut untuk hal tersebut. Jika anda mengalami kesulitan memahami produk reagen real time PCR, silahkan hubungi kontak kami atau konsultasi via WhatsApp : +6287777277740
Spesifikasi Real Time PCR
PT. Andaru Persada Mandiri sebagai distributor alat laboratorium menjual Real Time PCR(Polymerase Chain Reaction) dan berbagai instrumen laboratorium lainnya.
Perlu anda ketahui kami menawarkan harga PCR paling kompetitif. Kisaran harga PCR yang kami berikan berada di Rp. 615.000.000 (enam ratus lima belas jutaan). Harga ini (tidak bersifat mutlak)belum termasuk diskon jika anda membeli produk di bulan Juli 2020 ini.
Karena kami memberikan solusi terintergrasi, kami juga memberikan kepada anda :
- Diskusi(gratis) sebelum pembelian
- Installasi alat
- Training penggunaan awal
- Maintenance
- Service
- Garansi hingga 5 tahun.
- Leading Pettier Elements from MARLOW,ensure long life of 1,000,000 thermal cycles and fast ramping rate up to 6℃/S。
- T-opticalTM technology,reduce background noise,improve fluorescence signal sensitlvity and singnal to noise ratio.
- The angle of display could be adjusted to the best view.
- Simultaneous detection of wells,not in sequence.
- Users could view qPCR process and run PCR protocol through self-contained 10″TFT LCD and touch screen.
- Innovative SSLPTM CCD imaging technology for qPCR,avoiding more moving parts problems like overheat,wear and off center.Not optical fiber based, avoiding break and block by dust.
- Long life LED lamps to excite fluorescence ,no need for replacement in the future.
- Sample wells with temperature gradient function, convenient to optimize PCR conditions
- Equipped with LongGene’s “Optimun qPCR Design& Analysis Software” for remote operation of instruments and analysis of results.
- The drawer design of sample block,makes it easier to plck and place PCR tubes and plates
- LongGene’s “Optimun qPCR Design& Analysis Software” could be upgraded for free.
Instrument Performance
Sample Block Capacity : 96wells*0.1ml
Reaction Volume : 10-50ul
Tubes Option : White or clear PCR tubes or strips or 96well PCR piate,with optical flat cap
Heating&Cooling Technology : Peltier technonlong allow1,000,000 thermal cycles
Control Methods : Operated via PC or self-contaiend touch screen on instrument
Language : Englisn
Communications : USB2.0or LAN
Display : 10″Colcr TFT LCD and Touch Screen,view angle adjustable
Max.Number of programs : Max.15,000 programs onboard,unlimited storage of pretocols with USB flash drive
Fluorescence Detection
Excitation : Long life LED lanps
Detection : CCDs
Dynamic Range : 1-1010
Sensitivity : ≥1 copy
Calibrated Dyes at installation :
- F1:FAM, SYBRGreen
- F2:VIC, HEX, JOECY3, NED
- F3:ROX, TEXAS-RED
- F4 :CY5
- F5:CY5.5
- F6:Reserved
Fluorescence Excitation Range : 300-800nm
Fluorescence Detection Range : 500-800nm
Date Export Formats : Excel, TXT
Other Features
AC Power Supply : 100-240V,50-60Hz
Consupmtion : 600W
Net Weight : 13kg
Dimension(L*W*H) : 334*280*365mm
Computer Operating Systems : Windows10, Windows7, WindowsXP
Kontak PT. Andaru Persada Mandiri
Beberapa diantara anda mungkin belum mendapatkan informasi yang cukup mengenai reagen PCR maupun alat Real Time PCR yang sesuai.
Untuk itu silahkan hubungi kami sekarang, untuk mendapat konsultasi secara gratis sebelum melakukan pembelian.
PT. Andaru Persada Mandiri
Villa Bogor Indah 2, Blok BB2 No. 6, Kedunghalang, Bogor
WhatsApp & Hotline : 087777277740
Email : info.andarupm@gmail.com